Steam is capable of transferring a large amount of heat to materials used in food and beverage processing, and it can be precisely controlled to within narrow temperature ranges. It can be applied directly to the materials being heated (through Direct Steam Injection or DSI), or it can be applied indirectly through the use of heat exchangers that incorporate plates or tubes to segregate steam and process fluid. Depending on the application, there are advantages and disadvantages associated with each technique. The various applications for steam heating include production of hot water for sanitation, pasteurizing, and CIP, inline cooking of slurry type food products, drying and evaporating.
The amount of steam needed for these operations can be easily calculated. For heating a product in batch mode, the material (usually a liquid) is placed in a steam jacketed tank. The average heat transfer rate for the steam is governed by the fundamental conduction heat transfer equation:
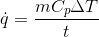
Where:
q̇ is the average heat transfer rate (kJ/sec)
m is the mass of the material that is being heated (kg)
Cp is the specific heat of the material to be heated (kJ/kg oC)
∆T is the required increase in temperature of the material to be heated (oC)
t is the total time over which heating occurs (sec)
The mass (m), temperature (∆T) and time (t) are process parameters, and value(s) of specific heat (Cp) can be obtained from tables for common liquids or solids. For reference, the specific heat of fresh water is 4.19 kJ/kg-oC (or 1 BTU/lb-oF). Heat transfer is dependent on ∆T across the jacket and effective surface area. Transfer is greatest at the start of the heating cycle, steadily diminishing as product temperature increases. When selecting the steam flow control valve and trap, both extremes must be considered.
For heating of a material that is flowing, the heat transfer equation becomes:
![]()
Where ṁ is the mass flow rate of the material (kg/sec).
Knowing the heat transfer rate (q̇) it is possible to calculate the mass of steam needed by:
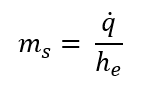
Where ms is the required mass flow rate of steam (kg/sec)
he is the steam evaporation energy or latent heat (kJ/kg), when using an indirect exchanger
he is the total steam energy - energy of inlet fluid when using a DSI heater
For a given temperature and pressure of steam, the evaporation energy and total energy (he) can be obtained from a saturated steam table. For example, 8 Barg saturated steam has an evaporation energy or latent heat content of 2033 kJ/kg and total heat content of 2773 kJ/kg. Once the required mass flow rate of (ms) the steam is known, it will be relatively straightforward to select the range of steam heating products that could be employed from a catalog, based on the specific application.
The difference in usable heat is one of the advantages of Direct Steam Injection (DSI). Following condensation, heat transfer continues until temperature equilibrium is reached between the condensate and product. When using a heat exchanger, only latent heat is transferred. Condensate is rapidly expelled before any further transfer can occur. Heat transfer is limited by average ∆T between steam and product and effective surface area. Transfer can be increased by increasing steam pressure and temperature, but at the cost of increased flash losses and decreased thermal efficiency.

Pick Heaters is the originator of Direct Steam Injection Heaters, having developed and patented a unique DSI system in 1945. Over a 80-year history, Pick Heaters has been an industry leader by continuously refining and innovating numerous DSI products in several industries, ranging from food to chemical and pharmaceutical processing, to pulp and paper, and textile to power plants.
For more information - contact us.